Study: Raloxifene prevents breast cancer as well as old standby, but with fewer side effects
WASHINGTON (AP) _ Women at high risk of breast cancer after menopause often shun a protective drug for fear of side effects. Now a striking study shows a widely used bone drug prevents breast cancer, tooTuesday, April 18th 2006, 8:26 am
By:
News On 6
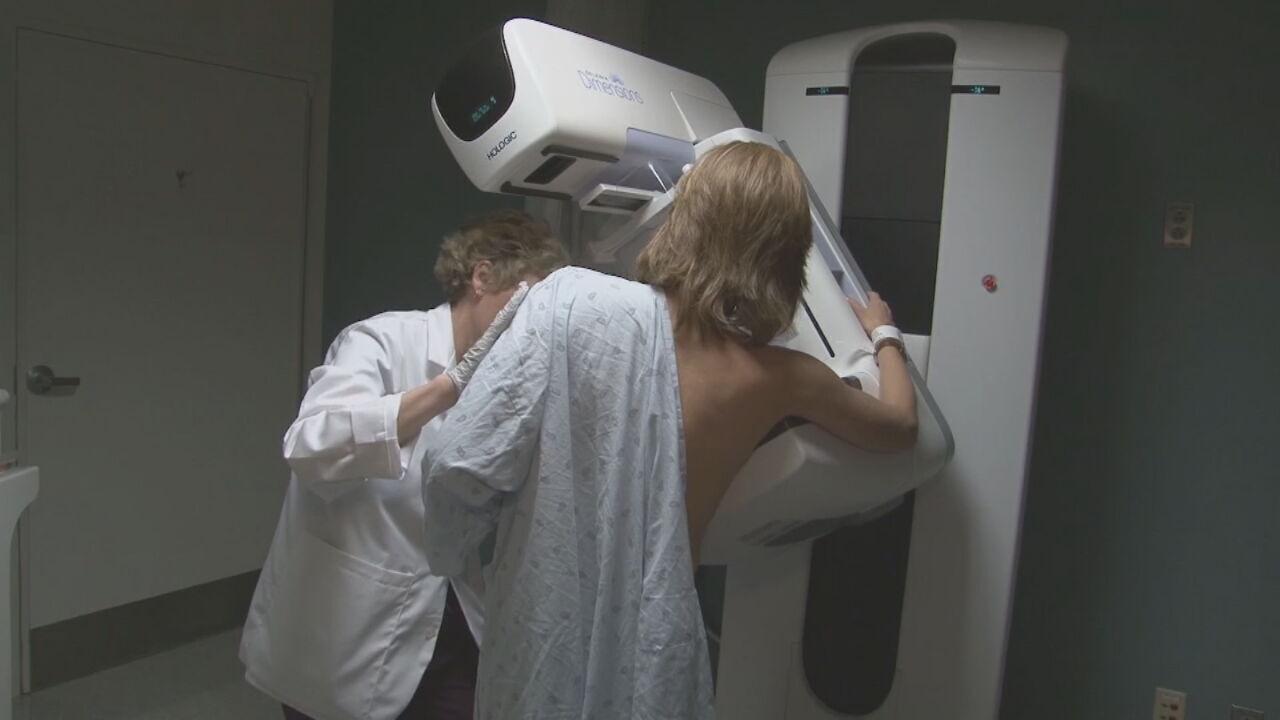
WASHINGTON (AP) _ Women at high risk of breast cancer after menopause often shun a protective drug for fear of side effects. Now a striking study shows a widely used bone drug prevents breast cancer, too _ a safer yet equally effective alternative to that old standby tamoxifen.
At least 2 million women might benefit from raloxifene's cancer-reducing effects, researchers said Monday in announcing initial results of the $88 million government study.
``Now women have a choice,'' Dr. Leslie Ford, NCI's cancer prevention chief, said in an interview. ``It's good news, because we're giving you a choice with fewer side effects.''
Until now, the decades-old tamoxifen has been doctor's only choice to reduce the chances of breast cancer striking high-risk women.
Manufacturer Eli Lilly & Co. already sells raloxifene, under the brand name Evista, to combat bone-thinning osteoporosis, another disease common after menopause. Armed with the new study of nearly 20,000 women, Lilly now plans to seek Food and Drug Administration approval to market raloxifene as a cancer-preventer, too.
Dr. Patrick Borgen, chief of breast service at Memorial Sloan-Kettering Cancer Center's surgery department, said Tuesday that raloxifene was now ``likely to become a primary weapon in reducing a post-menopausal woman's chance of developing breast cancer.''
But although doctors and patients may be tempted to start using raloxifene as a breast cancer prevention tool right away, they should wait until FDA approves the drug for that purpose, which should take only a few months, he said.
``I would advise them to wait until the FDA vets this data,'' Borgen said on ABC's ``Good Morning America.'' ``That won't be a long process.''
Both tamoxifen and raloxifene are so-called ``selective estrogen response modulators'' _ they act like the estrogen hormone in some tissues, but like an anti-estrogen in others. Estrogen can fuel certain breast cancers. But by acting like an estrogen in the uterus and bloodstream, tamoxifen causes some rare but serious side effects, increasing users' risk of getting uterine cancer or a life-threatening blood clot.
Raloxifene is a close chemical relative. The NCI study compared the two, and found that taking either tamoxifen or raloxifene daily for up to five years cut in half women's chances of developing invasive breast cancer.
Raloxifene causes the same side effects as tamoxifen, but not as many: Raloxifene users had 36 percent fewer uterine cancers and 29 percent fewer blood clots, the NCI announced Monday. Raloxifene users also suffered fewer vision-blocking cataracts.
While the reduction in those side effects was significant, the study also shows how uncommon they are. Thirty-six tamoxifen users developed uterine cancers, compared with 23 raloxifene users. The risk of blood clots was similarly low: 54 tamoxifen users had one in the lung, compared with 35 raloxifene users.
Still, many candidates for tamoxifen risk-reduction therapy have long avoided it for fear of those side effects, noted Dr. Kathy Albain of Loyola University, a study researcher.
``Here we have something that's a little less scary,'' Albain said. ``It might tip the scales for a lot of women.''
The new study means no change for pre-menopausal women _ there's no data showing whether raloxifene is safe for them, Albain stressed.
Nor does it mean that tamoxifen users should necessarily switch, she said. Women currently are prescribed tamoxifen for five years, and its breast cancer prevention benefit continues even after they stop taking the drug _ as raloxifene's seems to. So a woman already in, say, year 4 of her tamoxifen course with no sign of side effects probably has little to gain by switching, she explained.
But that's a question researchers were girding for as they spent Monday notifying study participants of the results.
One puzzle: While raloxifene was equally effective in blocking invasive breast cancer, it didn't protect quite as well as tamoxifen against noninvasive types of breast cancer such as ductal carcinoma in situ, noted Dr. Len Lichtenfeld of the American Cancer Society.
That type of tumor isn't life-threatening and shouldn't water down the overall message of raloxifene's benefit, said Dr. Victor Vogel of the University of Pittsburgh, who oversaw the study's design.
Among postmenopausal women, who's at high risk? Most of the study participants had a 4 percent chance of getting breast cancer within five years _ because of advanced age, a close relative with the disease, never having a child or having one late in life, or other well-known risk factors that women can calculate on a government Web site: http://cancer.gov/bcrisktool.
In simpler terms, for every 1,000 of those women, doctors expected 40 to develop breast cancer within five years if they did nothing, but taking one of the drugs cut that number to 20, Ford explained.
More Like This
April 18th, 2006
April 15th, 2024
April 12th, 2024
March 14th, 2024
Top Headlines
May 10th, 2024
May 10th, 2024